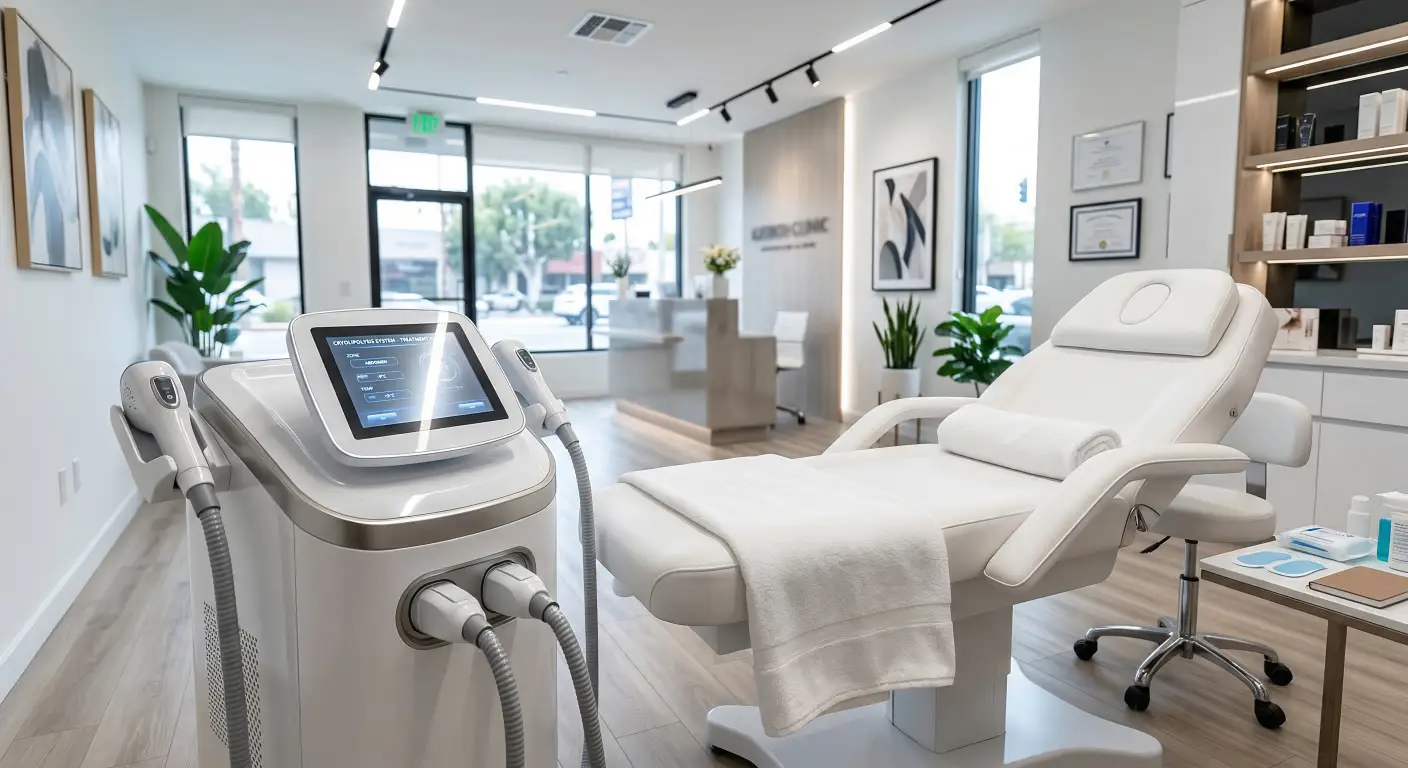
- 1. What are the key factors to consider when selecting a professional RF machine for skin tightening treatments?
- 2. How can I verify if a professional RF machine has FDA clearance and CE marking?
- 3. What are the potential risks of using a professional RF machine without proper FDA or CE certification?
- 4. How do I ensure that a professional RF machine complies with electromagnetic compatibility (EMC) standards?
- 5. What are the differences between FDA clearance and CE marking for professional RF machines?
- 6. How can I stay updated on regulatory changes affecting professional RF machines?
How to ensure FDA/CE compliance for the best professional rf machine?
1. What are the key factors to consider when selecting a professional RF machine for skin tightening treatments?
When selecting a professional RF machine for skin tightening, consider the following factors:
Technology Integration: Opt for devices that combine RF with other modalities like ultrasound or micro-needle technology to enhance treatment efficacy. For instance, the Alma Accent Prime integrates RF and ultrasound for comprehensive face and body treatments.
Safety Features: Ensure the machine includes advanced safety protocols such as real-time temperature monitoring and automatic shut-off mechanisms to prevent overheating.
Treatment Versatility: Choose machines that offer multiple applicators and adjustable energy levels to cater to various skin types and treatment areas.
Brand Reputation: Select devices from established manufacturers known for quality and reliability, as they are more likely to provide ongoing support and updates.
Regulatory Compliance: Verify that the machine meets international safety standards, including FDA clearance and CE marking, to ensure it adheres to necessary safety and performance criteria.
2. How can I verify if a professional RF machine has FDA clearance and CE marking?
To verify FDA clearance and CE marking for a professional RF machine:
FDA Clearance: Check the FDA's official database for medical devices to confirm if the device has been cleared for use in the United States.
CE Marking: Review the manufacturer's documentation or contact them directly to obtain proof of CE marking, indicating compliance with European Union health, safety, and environmental protection standards.
Manufacturer's Documentation: Request official certification documents from the manufacturer to ensure authenticity.
Third-Party Verification: Consult with regulatory bodies or industry experts to validate the device's compliance status.
3. What are the potential risks of using a professional RF machine without proper FDA or CE certification?
Using a professional RF machine without proper FDA or CE certification can lead to several risks:
Safety Hazards: Uncertified devices may lack essential safety features, increasing the risk of burns, skin damage, or other adverse effects.
Legal Implications: Operating uncertified equipment can result in legal consequences, including fines or business closure, as it may violate regulatory requirements.
Liability Issues: In the event of patient injury, using uncertified equipment can expose practitioners to liability claims.
Insurance Complications: Insurance providers may refuse coverage for treatments performed with uncertified devices, leading to financial losses.
4. How do I ensure that a professional RF machine complies with electromagnetic compatibility (EMC) standards?
To ensure compliance with EMC standards for a professional RF machine:
Review Technical Documentation: Examine the manufacturer's technical files to confirm that the device meets EMC requirements.
Request Certification Reports: Obtain EMC certification reports from the manufacturer or a recognized testing laboratory.
Consult Regulatory Guidelines: Refer to guidelines provided by regulatory bodies, such as the FDA's recommendations for EMC in healthcare facilities, to understand compliance expectations.
Conduct Independent Testing: If necessary, arrange for independent EMC testing to verify the device's compliance.
5. What are the differences between FDA clearance and CE marking for professional RF machines?
The differences between FDA clearance and CE marking include:
Regulatory Scope:
- FDA Clearance: Applies to devices intended for the U.S. market, ensuring they meet U.S. safety and effectiveness standards.
- CE Marking: Applies to devices intended for the European market, indicating compliance with EU health, safety, and environmental protection standards.
Certification Process:
- FDA Clearance: Involves a premarket notification process where manufacturers must demonstrate that their device is substantially equivalent to a legally marketed device.
- CE Marking: Often a self-declaration process, where manufacturers assess compliance and compile technical documentation, engaging a Notified Body only if the device meets certain risk thresholds.
Market Authorization:
- FDA Clearance: Required for devices marketed in the U.S.
- CE Marking: Required for devices marketed in the EU.
6. How can I stay updated on regulatory changes affecting professional RF machines?
To stay updated on regulatory changes affecting professional RF machines:
Monitor Regulatory Websites: Regularly check official websites of regulatory bodies such as the FDA and the European Medicines Agency (EMA) for updates.
Subscribe to Industry Newsletters: Join newsletters from reputable industry organizations and associations that provide timely information on regulatory changes.
Attend Industry Conferences: Participate in conferences and seminars where regulatory experts discuss recent developments.
Consult Regulatory Consultants: Engage with consultants who specialize in medical device regulations to receive personalized guidance.
In conclusion, selecting a professional RF machine requires careful consideration of technology integration, safety features, treatment versatility, brand reputation, and regulatory compliance. Ensuring that the device has proper FDA clearance and CE marking is crucial for legal operation and patient safety. Staying informed about regulatory changes is essential for maintaining compliance and delivering effective treatments. For personalized assistance and to explore our range of FDA-cleared and CE-marked professional RF machines, please contact us at coco@huimainbeauty.com.



HMR-1 台式磁立环
Who can use this Magnetic Conduction Ring machine?
It is suitable for beauty salons, aesthetic clinics, physical therapy centers, and rehabilitation facilities.
HMT 42 R
How many treatments are needed to see results?
Most clients begin to notice visible changes after 4–6 sessions, depending on their body type and treatment goals.
OL-153
What skin types can the Hydro Dermabrasion Water Oxygen Machine treat?
It’s suitable for all skin types, including sensitive and dry skin, offering deep hydration and exfoliation.
台式欧洲之星
Is this CO2 fractional laser safe for all skin types?
Yes, but it is most effective for light to medium skin tones. Darker skin types should be treated with caution.
HMT82-2R
Who can benefit from this device?
It is designed for beauty salons, weight loss clinics, fitness centers, and distributors who want to expand their service offerings.




Get more information
If you have any comments or good suggestions, please leave us a message; later our professional staff will contact you as soon as possible.
© 2025 HUIMAIN. All Rights Reserved. Privacy Policy | Terms and Conditions | Sitemap




Facebook
Instagram
Medical beauty equipment factory
huimain Medical beauty equipment factory
beauty machine supplier
Whatsapp: +8619124004543